similarities and trends
Melting point, ionization energy, atomic radius, chemical reactivity, ion charge and conductivity are properties of elements that show similarities and trends among the period table.
MELTING POINT
TRENDS and SIMILARITIES:
- melting points and molecular mass of binary carbon-halogen compounds and hydrogen halides are due to intermolecular forces
- melting destroys the arrangement of atoms in a solid, making the amount of heat needed for melting depend on the strength of attraction between atoms
- strength of attraction increases as the number of electrons increase
- increase in electrons increases bonding
- heat and electricity conductibility vary through out
EXAMPLE:
Melting point of HF should be approximately - 145 ̊C based off melting points of HCl, HBr, and HI, but the observed value is -83.6̊ (Petrucci)
- melting points and molecular mass of binary carbon-halogen compounds and hydrogen halides are due to intermolecular forces
- melting destroys the arrangement of atoms in a solid, making the amount of heat needed for melting depend on the strength of attraction between atoms
- strength of attraction increases as the number of electrons increase
- increase in electrons increases bonding
- heat and electricity conductibility vary through out
EXAMPLE:
Melting point of HF should be approximately - 145 ̊C based off melting points of HCl, HBr, and HI, but the observed value is -83.6̊ (Petrucci)
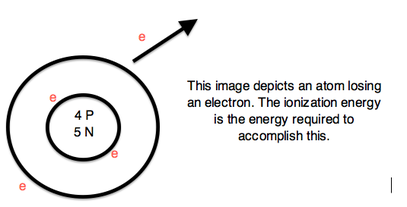
IONIZATION ENERGY
|
- the energy required to completely remove an electron from a gaseous atom or ion
- Ionization Energy is always positive TRENDS and SIMILARITIES: - energy required to remove one valence electron is the first ionization energy - second ionization energy is the energy required to remove a second valence electron, and so on EXAMPLES: 1st ionization energy equation X → X+ + e- 2nd ionization energy equation X+ → X2+ + e- 3rd ionization energy equation X2+ → X3+ + e- - highest ionization energies are the noble gases because they all have high effective charge due to their octet formation and require a high amount of energy to destroy that stable configuration - highest energy required occurs with elements in the upper right hand corner - elements in left hand corner have low ionization energy because losing an electron allows them to have the noble gas configuration. Therefore, it requires less energy to remove one of there valence electrons - ionization energy decreases down a group - ionization energy increases across a period |
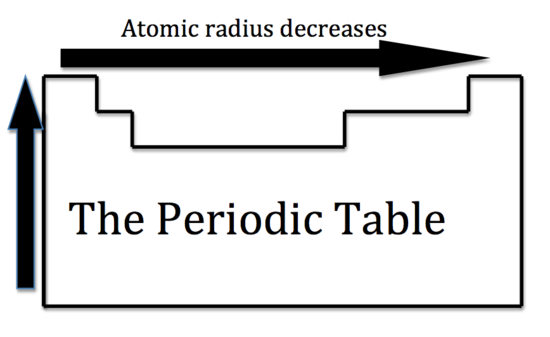
ATOMIC RADIUS
|
- we can never determine the atomic radius of an atom because there is never a zero probability of finding an electron
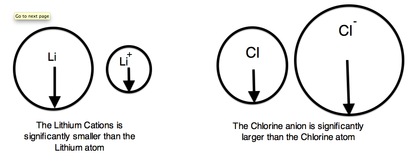
- we can measure the distance between two radii TRENDS and SIMILARITIES: -The atomic number increases moving left to right across a period and subsequently so does the effective nuclear charge. Therefore, moving left to right across a period the nucleus has a greater pull on the outer electrons and the atomic radii decreases. Moving down a group in the periodic table, the number of filled electron shells increases. In a group, the valence electrons keep the same effective nuclear charge, but now the orbitals are farther from the nucleus. Therefore, the nucleus has less of a pull on the outer electrons and the atomic radii are larger. - cation is an atom that has lost one of its outer electrons - cations have a smaller radius than the atom that they were formed from. - With the loss of an electron, the positive nuclear charge out powers the negative charge that the electrons exert. The positive nucleus pulls the electrons tighter and the radius is smaller. An anion is an atom that has gained an outer electron. - Anions have a greater radius than the atom that they were formed from. The gain of an electron does not alter the nuclear charge, but the addition of an electron causes a decrease in the effective nuclear charge. Therefore, the electrons are held more loosely and the atomic radius is increased. - atomic radius increases as you go down a group - atomic radius decreases as you go across a period |
CHEMICAL REACTIVITY
TRENDS and SIMILARITIES:
TRENDS and SIMILARITIES:
IN METALS!
- reactivity increases as you go down a group because the farther down a group of metals you go, the easier it is for electrons to be given or taken away, resulting in higher reactivity.
- reactivity decreases as you go across a period because though they still want to give away valence electrons they have more of them to get rid of, which requires more energy. Not as easy to blow off a little steam!
IN NON-METALS!
- reactivity increases as you go up a group because the higher up and to the right atoms are, the higher the electronegativity, resulting in a more vigorous exchange of electrons. Fluorine? A greedy, impatient beast when it comes to electron exchange manners.
- reactivity increases as you go across a period because the closer you get to fulling your s- and p- orbitals the more motivated you are to do so.
(notice how trends repeat?)
- reactivity increases as you go down a group because the farther down a group of metals you go, the easier it is for electrons to be given or taken away, resulting in higher reactivity.
- reactivity decreases as you go across a period because though they still want to give away valence electrons they have more of them to get rid of, which requires more energy. Not as easy to blow off a little steam!
IN NON-METALS!
- reactivity increases as you go up a group because the higher up and to the right atoms are, the higher the electronegativity, resulting in a more vigorous exchange of electrons. Fluorine? A greedy, impatient beast when it comes to electron exchange manners.
- reactivity increases as you go across a period because the closer you get to fulling your s- and p- orbitals the more motivated you are to do so.
(notice how trends repeat?)
CHECK OUT THESE TWO AWESOME VIDEOS
ION CHARGE and CONDUCTIVITY
TRENDS and SIMILARITIES:
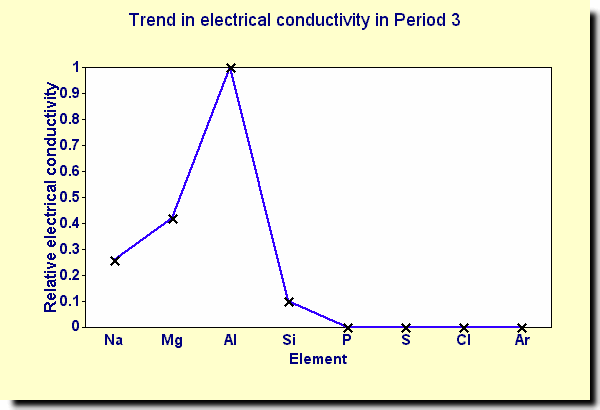
Conductivity :
Let's take a look at the conductivity trends in period 3
Conductivity :
Let's take a look at the conductivity trends in period 3
-For an element to conduct electricity, it must contain electrons that are free to move. In general, metals are good conductors of electricity and non-metals are poor conductors of electricity.
SO WHY EXACTLY ARE THERE DIFFERENT CONDUCTIVITY LEVELS?
Sodium, magnesium and aluminium are all metals, they have metallic bonding
SO WHY EXACTLY ARE THERE DIFFERENT CONDUCTIVITY LEVELS?
Sodium, magnesium and aluminium are all metals, they have metallic bonding
- - the number of delocalised electrons increases ...
- - there are more electrons which can move and carry charge ...
- - so the electrical conductivity increases.
- AND WHAT ABOUT THE REST?
- The remaining elements in Period 3 do not conduct electricity because they are all Non-Metals
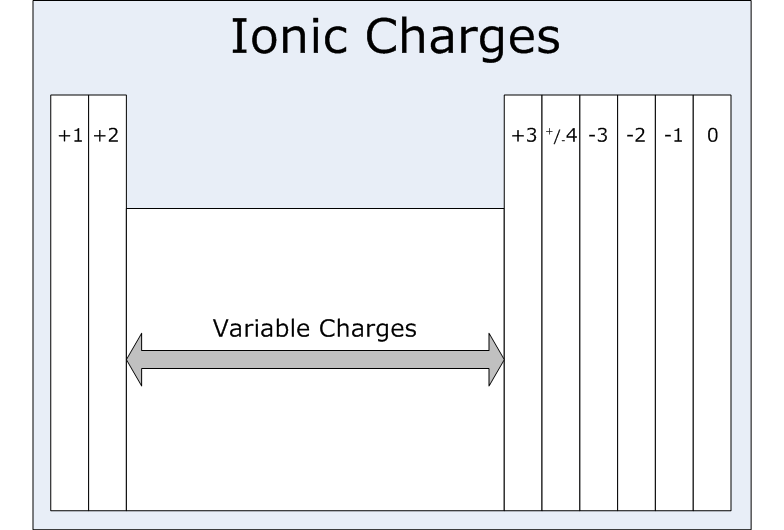
ION CHARGE:
- The number of electrons an element tends to gain or lose is a periodic property. The alkali metals always form +1 ions. Hydrogen can form either +1 or -1 ions. The alkaline earth metals always form +2 ions. Nitrogen and phosphorus in group 15 usually form -3 ions. The chalcogens usually form -2 ions. The halogens always form -1 ions. The noble gases don’t readily form ions. Many of the transition metals form more than one type of ion.
- This is needed for formula naming and solving
- The number of electrons an element tends to gain or lose is a periodic property. The alkali metals always form +1 ions. Hydrogen can form either +1 or -1 ions. The alkaline earth metals always form +2 ions. Nitrogen and phosphorus in group 15 usually form -3 ions. The chalcogens usually form -2 ions. The halogens always form -1 ions. The noble gases don’t readily form ions. Many of the transition metals form more than one type of ion.
- This is needed for formula naming and solving
THIS PICTURE SHOULD HELP YOU UNDERSTAND