classification
Elements of the periodic table can be classified in many different ways such as metalloids, metals and non-metals.
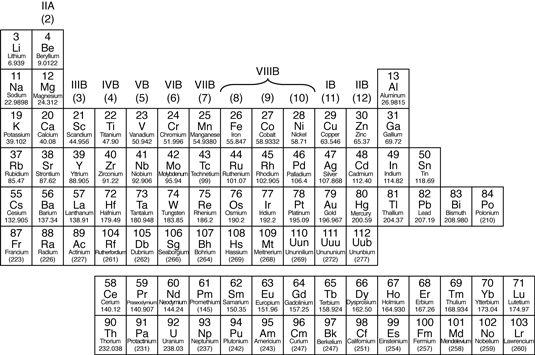
mETALS
In the periodic table, you can see a stair-stepped line starting at Boron (B),and going all the way down to Polonium (Po), Except for Germanium (Ge) and Antimony (Sb), all the elements to the left of that line can be classified as metals.
These metals have properties that you normally associate with the metals you encounter in everyday life:
These metals have properties that you normally associate with the metals you encounter in everyday life:
- usually solid at room temperature (mercury is an exception)
- high luster (shiny)
- metallic appearance
- good conductors of heat and electricity
- malleable (can be bent and pounded into thin sheets)
- ductile (can be drawn into wire)
- corrode or oxidize in air and sea water
- usually dense (exceptions include lithium, potassium, and sodium)
- may have very high melting point
- readily lose electrons
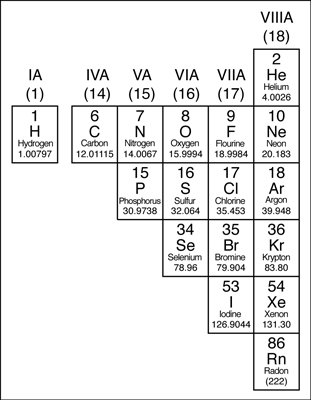
non-metals
Except for the elements that border the stair-stepped line, the elements to the right of the line are classified as nonmetals (along with hydrogen). Nonmetals have properties opposite those of the metals.
The nonmetals are brittle, not malleable or ductile, poor conductors of both heat and electricity, and tend to gain electrons in chemical reactions. Some nonmetals are liquids.
The nonmetals are brittle, not malleable or ductile, poor conductors of both heat and electricity, and tend to gain electrons in chemical reactions. Some nonmetals are liquids.
- dull appearance
- usually brittle
- poor conductors of heat and electricity
- usually less dense, compared to metals
- usually low melting point of solids, compared with metals
- tend to gain electrons in chemical reactions
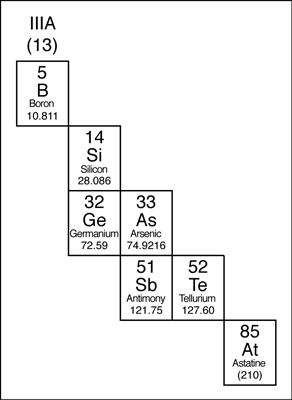
MetAlloids
The elements that border the stair-stepped line are classified as metalloids. The metalloids, or semimetals, have properties that are somewhat of a cross between metals and nonmetals.
Metalloids tend to be economically important because of their unique conductivity properties (they only partially conduct electricity), which make them valuable in the semiconductor and computer chip industry. The metalloids are shown in the following illustration.
Metalloids tend to be economically important because of their unique conductivity properties (they only partially conduct electricity), which make them valuable in the semiconductor and computer chip industry. The metalloids are shown in the following illustration.
- dull or shiny
- usually conduct heat and electricity, though not as well as metals
- often make good semiconductors
- often exist in several forms
- often ductile
- often malleable
- may gain or lose electrons in reactions